incomplete octet of electrons|incomplete octet examples : Cebu The second exception to the Octet Rule is when there are too few valence . Book New Coast Hotel Manila, Manila on Tripadvisor: See 3,503 traveler reviews, 2,134 candid photos, and great deals for New Coast Hotel Manila, ranked #53 of 1,271 hotels in Manila and rated 4 of 5 at Tripadvisor. . Car hire. Taxi service. Business Center with Internet Access. Conference facilities. Banquet room. Meeting rooms. Spa. Massage .
PH0 · octet rule violation
PH1 · incomplete octet examples
PH2 · boron octet rule exception
PH3 · Iba pa
Jinx swaps weapons between Fishbones the rocket launcher and Pow-Pow the minigun. While using the rocket launcher, Jinx's attacks deal 110% of attack damage as physical damage to the target and nearby enemies, gains 80 / 110 / 140 / 170 / 200 attack range, costs 20 mana per rocket and scales 10% less with bonus attack speed.
incomplete octet of electrons*******The second exception to the Octet Rule is when there are too few valence electrons that results in an incomplete Octet. There are even more occasions where the octet rule does not give the most correct depiction of a molecule or ion.
The second exception to the Octet Rule is when there are too few valence .
The second exception to the Octet Rule is when there are too few valence .
The second exception to the Octet Rule is when there are too few valence electrons that results in an incomplete Octet. There are even more occasions where .Incomplete Octet. There are certain atoms of certain elements that can exist in stable compounds forming bonds with less than eight valence electrons. When this occurs, the .The octet rule states that atoms with an atomic number below 20 tend to combine so that they each have eight electrons in their valence shells, which gives them the same .
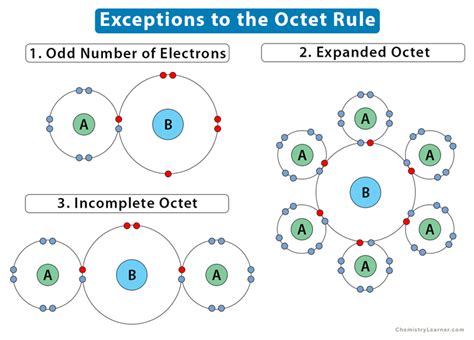
While most atoms obey the duet and octet rules, there are some exceptions. For example, elements such as boron or beryllium often form compounds in which the .incomplete octet of electrons While Lewis electron dot structures help determine bonding in most compounds, there are three general exceptions: molecules in which atoms have fewer than eight electrons (boron chloride and .1. Odd Number of Electrons. Usually, molecules containing s- and p-block elements have an even number of electrons (ex. H 2 O, CO 2, and NH 3 ). However, a few molecules .
Molecules with an Incomplete Octet on a Central Atom. Some molecules contain a central atom that does not have a filled valence shell. Usually, these central atoms are from .Exceptions to the Octet Rule. Many covalent molecules have central atoms that do not have eight electrons in their Lewis structures. These molecules fall into three categories: Odd .The second exception to the Octet Rule is when there are too few valence electrons that results in an incomplete Octet. There are even more occasions where the octet rule . Exceptions to the octet rule fall into one of three categories: (1) an incomplete octet, (2) odd-electron molecules, and (3) an expanded octet. Incomplete Octet. In some compounds, the number of electrons surrounding the central atom in a stable molecule is fewer than eight. Beryllium is an alkaline earth metal and so may be .incomplete octet of electrons incomplete octet examples Exceptions to the octet rule fall into one of three categories: (1) an incomplete octet, (2) odd-electron molecules, and (3) an expanded octet. Incomplete Octet. In some compounds, the number of electrons surrounding the central atom in a stable molecule is fewer than eight. Beryllium is an alkaline earth metal and so may be .The "octet rule" says that in many compounds the most stable (correct) electron configuration is when there are 8 electrons (four filled orbitals). This is a consequence of the fact that many compounds involve the s .incomplete octet examplesIncomplete Octet. There are certain atoms of certain elements that can exist in stable compounds forming bonds with less than eight valence electrons. When this occurs, the atom of the element within the molecule is said to contain an incomplete octet. The common examples of such elements are hydrogen (stable with only 2 valence electrons .

Incomplete Octet. Lighter s- and p-block elements can form compounds with less than 8 valence electrons and hence, have incomplete octets. Hydrogen, lithium, beryllium, and boron have fewer electrons to complete the octet. Helium has 2 electrons in its valence shell but is an inert gas. It does not form a bond with other atoms.
Exception 2: Incomplete Octets. The second exception to the Octet Rule is when there are too few valence electrons that results in an incomplete Octet. There are even more occasions where the octet rule does not give the most correct depiction of a molecule or ion. This is also the case with incomplete octets.
Exception 2: Incomplete Octets. The second exception to the Octet Rule is when there are too few valence electrons that results in an incomplete Octet. There are even more occasions where the octet rule does not give the most correct depiction of a molecule or ion. This is also the case with incomplete octets. Exception 2: Incomplete Octets. The second exception to the Octet Rule is when there are too few valence electrons that results in an incomplete Octet. There are even more occasions where the octet rule does not give the most correct depiction of a molecule or ion. This is also the case with incomplete octets.
Up-to-date list of no deposit bonus offers for online casinos in September 2024. Find best no deposit casino bonus codes and free spins bonuses.
incomplete octet of electrons|incomplete octet examples